Recalls / 2014-07-14
GSK Recalls Panadol Advance Bottles Due to Failure to Meet Child-Resistant Closure Requirement; Sold Exclusively in Puerto Rico
14229 · Recalled 2014-07-14 · CPSC page
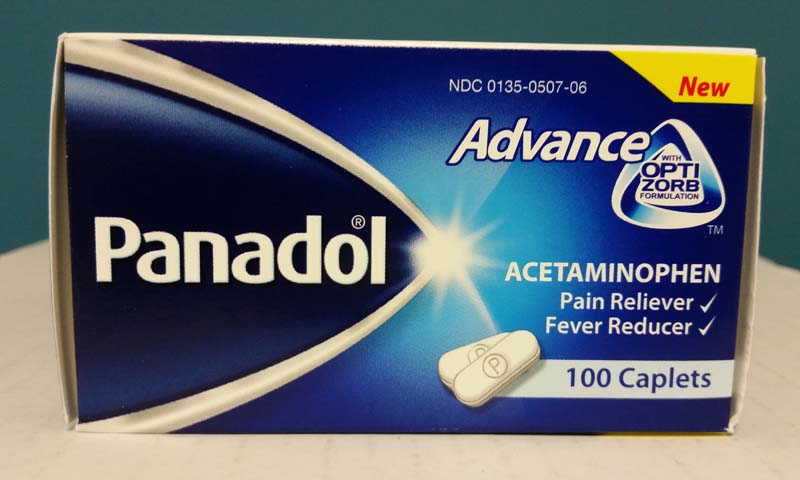
Description
This recall involves bottles of 100ct Panadol Advance pain relievers. The medicine was sold in white containers with a blue label, inside a blue box. "Panadol" and "Advance" are printed on the label. Lot numbers and dates codes are located on the left side panel of the box and on the left side of the label on the bottle, near the bar code. Lot numbers and date codes included in the recall are: Lot number: 14241, expiration date: 02/2015 Lot number: 14002, expiration date: 10/2014 Lot number: 13881, expiration date: 09/2014 Lot number: 13801, expiration date: 09/2014
Hazards
Reported injuries
Remedy
Products
| Name | Model | Units |
|---|---|---|
| Panadol Advance® 100 count caplets | About 10,600 |
Parties
- Manufacturer
- GlaxoSmithKline (GSK)
- Made in
- United States
- Sold at
- Drug, grocery, mass merchandise stores in Puerto Rico
Consumer contact
GlaxoSmithKline (GSK) toll-free at (888) 912-8455 from 9 a.m. to 5 p.m. ET Monday through Friday, or online at www.us.gsk.com and click on "Media and news" in the upper left and, if not listed with current releases, click on "Press release archive" on the left side for more information.
More images

Source: CPSC recall 14229. Inclusion in this database is not legal or medical advice.