Recalls / 2016-07-27
Novel Laboratories Recalls Zolpidem Tartrate Blister Packs Due to Failure to Meet Child-Resistant Closure Requirement; Risk of Poisoning
16232 · Recalled 2016-07-27 · CPSC page
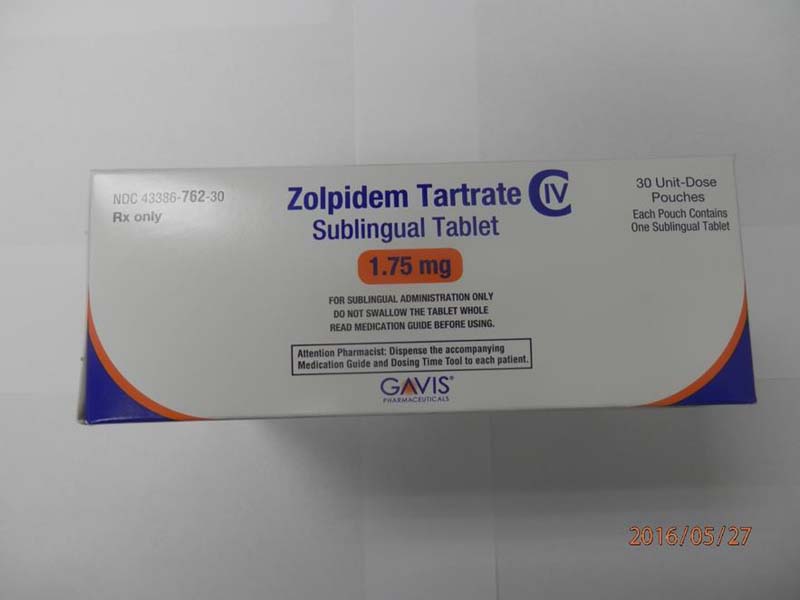
Description
This recall involves 1.75 mg and 3.5 mg Zolpidem Tartrate sublingual (rapid dissolve) tablets from Novel Laboratories. The prescription medication is a sleep drug. The packaging for the 1.75 mg tablets has lot number M16140A, 1.75 mg and national drug code (NDC) 43386-762-30. The packaging for the 3.5 mg tablets has lot number M16144A, 3.5mg and national drug code (NDC) 43386-761-30. The Zolpidem Tartrate is packaged as a single tablet in a peel-and-push blister pack inside an outer open-ended pouch with 30 pouches per box. The lot number and an expiration date of 02/2018 are printed on the bottom left of the pouches. The NDC is printed on the top left corner of the boxes. "Gavis" is printed in blue on the center of the white boxes and pouches.
Hazards
Reported injuries
Remedy
Products
| Name | Model | Units |
|---|---|---|
| Zolpidem Tartrate Sublingual tablets | About 5,700 boxes of 30 blister packs |
Parties
- Manufacturer
- Novel Laboratories
- Distributor
- Gavis Pharmaceuticals, of Somerset, N.J.
- Made in
- United States
- Sold at
- Clinics, pharmacies nationwide, including CVS, Costco, Walgreens, Kroger, Wegmans
Consumer contact
Novel Laboratories toll-free at (866) 403-7592 from 9 a.m. to 5 p.m. ET Monday through Friday or online at www.novellabs.net and click on "Important Zolpidem Information" for more information.
More images


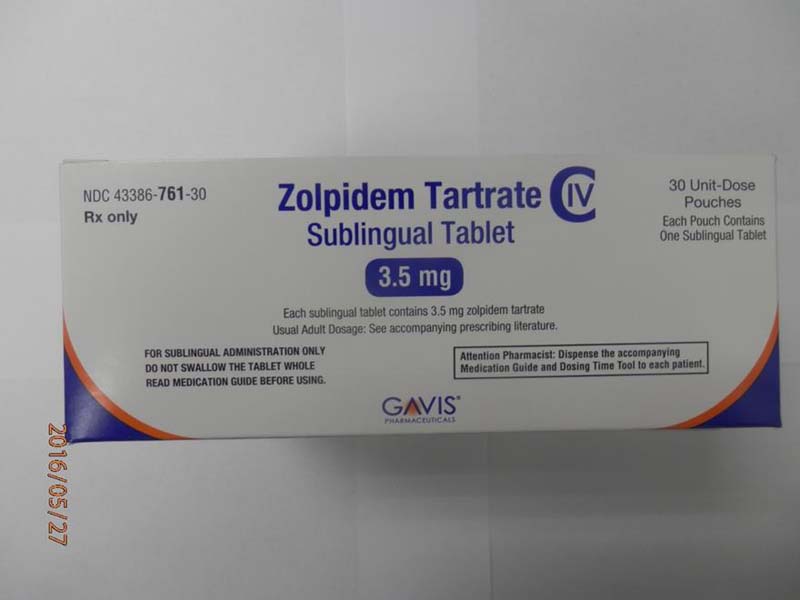
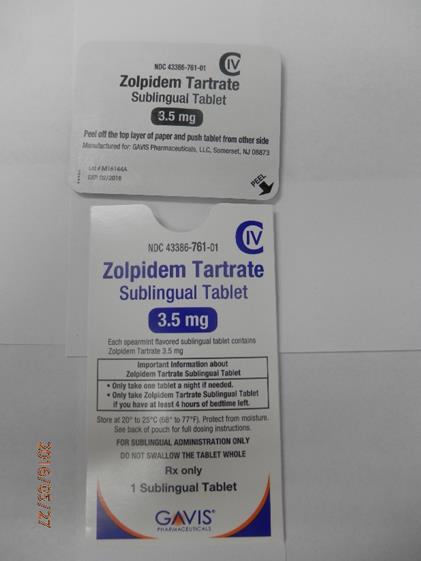

Source: CPSC recall 16232. Inclusion in this database is not legal or medical advice.