Recalls / 2018-12-19
Women's Iron Complete Supplements Recalled by GNC Due to Failure to Meet Child Resistant Closure Requirement; Risk of Poisoning
19055 · Recalled 2018-12-19 · CPSC page

Description
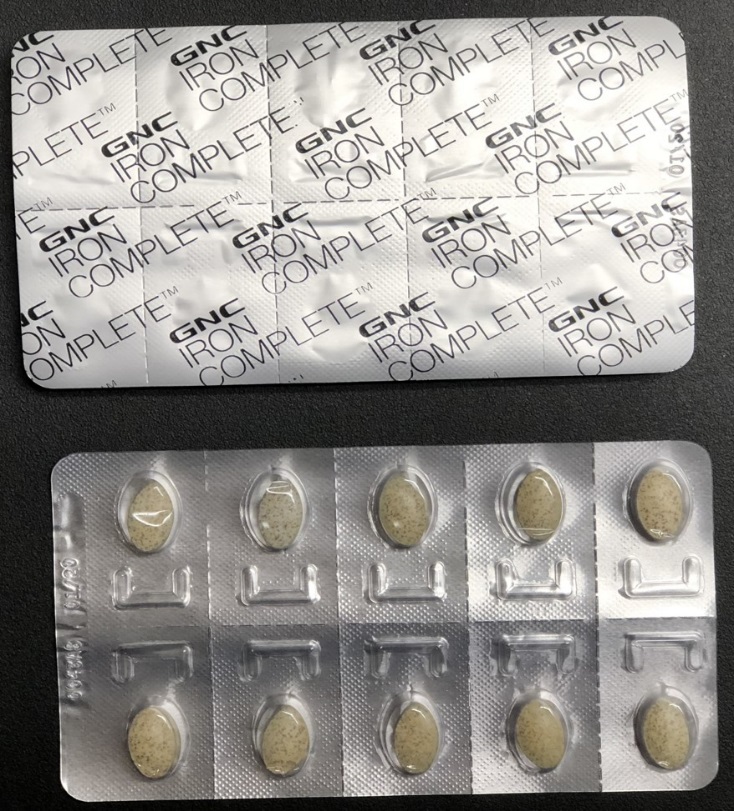
This recall involves Women's Iron Complete Dietary Supplement 60-count caplets. The recalled dietary supplement is in a white box with "Women's Iron Complete" printed on the front in gray and red font. The box contains blister packets with a total of sixty caplets.
Hazards
The dietary supplement blister packaging is not child resistant, as required by federal law. If ingested by a child, these supplements could cause serious injury or death.
Reported injuries
None reported
Remedy
Consumers should keep these products out of the reach of children and contact GNC for instructions on how to obtain a refund. Consumers can return the unused product to their local GNC store for a refund.
Products
| Name | Model | Units |
|---|---|---|
| Women's Iron Complete Dietary Supplement (60 caplets) | About 756,000 |
Parties
- Manufacturer
- Nutra Manufacturing, Inc.
- Distributor
- General Nutrition Corp., of Pittsburgh, Pa.
- Made in
- United States
- Sold at
- GNC retail, online at www.gnc.com, online at www.drugstore.com, through August 2016 for about $10.
Consumer contact
GNC at 888-462-2548 any time, email at customer-service@gnc-hq.com or online at www.gnc.com and click on “Recall Notice” link at the top of the page for more information.
More images
Source: CPSC recall 19055. Inclusion in this database is not legal or medical advice.