Recalls / 2019-12-11
Profile Design Recalls Bicycle Carbon Aerobars Due to Crash Hazard
20039 · Recalled 2019-12-11 · CPSC page

Description
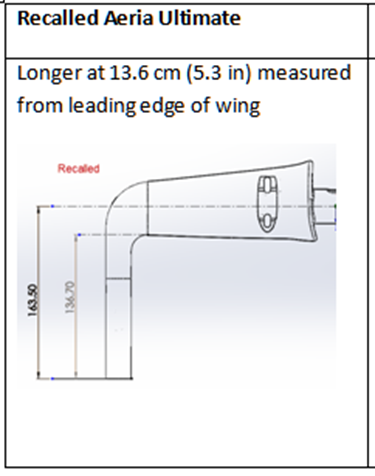
This recall involves Profile Design Aeria Ultimate carbon aerobars sold individually and as an original equipment upgrade on consumer configurable Dimond Brand Bikes and Quintana Roo model bicycles. The aerobars were sold in one size and are all matte black in color. "Profile Design" and "Ultimate" are embossed in gloss black on the top of the rear edge of the aerobars. The best way to identify recalled handlebars is to measure the uncut hand extensions from the leading edge of wing to tip of hand extension (where brake lever is placed). On recalled bars it is 13.6 cm (5.3 in) measured from leading edge of wing.
Hazards
Reported injuries
Remedy
Products
| Name | Model | Units |
|---|---|---|
| Aeria Ultimate carbon aerobars | About 260 |
Parties
- Importer
- Profile Design, of Carson, Calif.
- Made in
- Taiwan
- Sold at
- Independent bicycle from May 2017 through October 2018 for about $1, 300 for the aerobar sold individually or, 200, $14, 000 as original equipment on bicycles.
Consumer contact
Profile Design toll-free at 888-800-5999 from 9 a.m. to 5 p.m. PT Monday through Friday or online at www.profile-design.com and click on “Recall Notices” or email AeriaUltimateRecall@profile-design.com for more information.
More images

Source: CPSC recall 20039. Inclusion in this database is not legal or medical advice.