Recalls / 2020-05-27
Sanvall Enterprises Recalls Rapid Alivio Pain Relieving Roll-On Due to Failure to Meet Child Resistant Packaging Requirement; Risk of Poisoning
20126 · Recalled 2020-05-27 · CPSC page
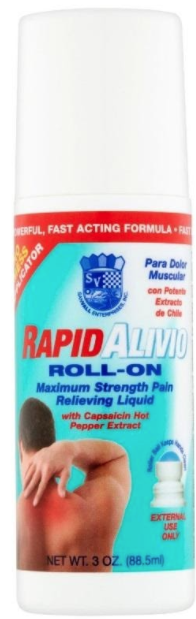
Description
This recall involves Sanvall Rapid Alivio Pain Relieving Roll-On in a 3 fl. oz (88.5 mL) white plastic bottle with a white cap and a red, white and blue label. "Rapid Alivio," "Maximum Strength Pain Relieving Liquid" and "Para Dolor Muscular" are printed on the label. Lot numbers 18032201, 18032301, 19040501, or 19052801 are printed on the bottom of the bottle. UPC code 605100014225 is printed on the side of the label.
Hazards
Reported injuries
Remedy
Products
| Name | Model | Units |
|---|---|---|
| Sanvall Rapid Alivio Pain Relieving Roll-On | 5400 |
Parties
- Manufacturer
- Sanvall Enterprises Inc.
- Made in
- United States
- Sold at
- Navarro Discount Pharmacy, Walmart stores in South Florida
Consumer contact
Sanvall Enterprises collect at 305-887-1090 from 9 a.m. to 5 p.m. ET Monday through Friday, email at SanarNaturals@Sanvall.com and in the body of the email provide your name, address, and photo of the product or online at www.Sanvall.com and click on “Recall - Important Safety Information - Rapid Alivio Roll-On” for more information.
More images


Source: CPSC recall 20126. Inclusion in this database is not legal or medical advice.