Recalls / 2020-03-18
NumbSkin Pain Relief Cream Recalled by SeeNext Venture Due to Failure to Meet Child Resistant Packaging Requirement; Risk of Poisoning (Recall Alert)
20724 · Recalled 2020-03-18 · CPSC page
Description
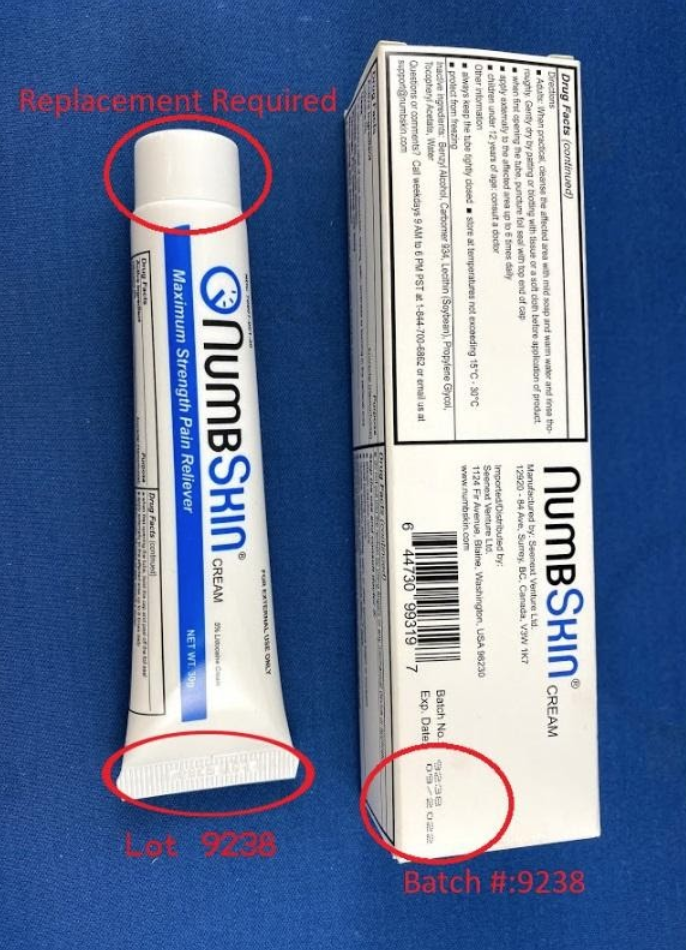
This recall involves NumbSkin pain relief cream with 5% lidocaine. The topical anesthetic cream was sold in 30 grams in a white with blue tube. NumbSkin is printed on the tube. Lot 9238 and a date code of 10/2019 through 09/2020 in a MM/YYYY format is embossed on the tub's thin end. Batch number 9238 is printed on the product packaging.
Hazards
The packaging is not child resistant as required by the Poison Prevention Packaging Act. The pain relieving cream contains lidocaine, posing a risk of poisoning to young children if they put it on their skin or ingest it.
Reported injuries
None reported.
Remedy
Consumers should immediately store the pain relief cream in a safe location out of reach of children and contact SeeNext Venture for instructions on how to dispose or return it and to receive a free replacement similar product with a child-resistant cap. Amazon is contacting all known purchasers directly.
Products
| Name | Model | Units |
|---|---|---|
| NumbSkin pain relief cream with lidocaine | About 8,000 |
Parties
- Importer
- SeeNext Venture Ltd., of Blaine, Wash.
- Made in
- Canada
- Sold at
- Online at Amazon.com
Consumer contact
SeeNext Venture toll-free at 844-700-6862 from 9 a.m. to 6 p.m. PT Monday through Saturday, email at distributions@numbskin.com or online at www.numbskin.com and click on Recall at the home page for more information.
Source: CPSC recall 20724. Inclusion in this database is not legal or medical advice.