Recalls / 2020-04-23
Natural Health Partners Recalls Dr. Mercola Wintergreen Essential Oils Due to Failure to Meet Child Resistant Packaging Requirement; Risk of Poisoning (Recall Alert)
20736 · Recalled 2020-04-23 · CPSC page

Description
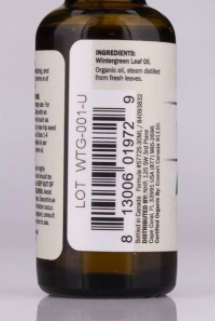
This recall involves Dr. Mercola Wintergreen Essential Oil in a 1 fl.oz (30 mL) amber glass bottles with black caps. The Lot codes for the recalled products are WTG-001-T and WTG-001-U, the UPC code is 813006019729. The Lot codes are printed on the product label above the UPC code.
Hazards
The products contain the substance methyl salicylate which must be in child resistant packaging as required by the Poison Prevention Packaging Act (PPPA). The packaging of the product is not child resistant, posing a risk of poisoning if the contents are swallowed by young children.
Reported injuries
None reported
Remedy
Consumers should store the product in a safe location out of reach of children and contact Natural Health Partners for a free replacement child resistant cap or a full refund. Natural Health Partners is contacting all known purchasers directly about the recall.
Products
| Name | Model | Units |
|---|---|---|
| Dr. Mercola Wintergreen Essential Oil | About 5,600 |
Parties
- Manufacturer
- LiquiFab
- Importer
- Atrium Innovations, of Pittsburgh, Pa.
- Distributor
- Natural Health Partners, of Cape Coral, Fla.
- Made in
- Canada
- Sold at
- Online at www.shop.mercola.com
Consumer contact
Natural Health Partners toll-free at 877-985-2696 from 8 a.m. to 9 p.m. ET Monday through Friday and 9 a.m. to 6 p.m. ET Saturday through Sunday, email at csmgt@mercola.com or online at www.shopmercola.com and click on ‘HELP’ at the top right corner of the page for more information.
More images
Source: CPSC recall 20736. Inclusion in this database is not legal or medical advice.