Recalls / 2025-09-11
Blueroot Health Recalls Bottles of Bariatric Fusion Iron Multivitamins Due to Risk of Serious Injury or Death from Child Poisoning; Violates Mandatory Standard for Child-Resistant Packaging; Manufactured by VitaQuest International
25460 · Recalled 2025-09-11 · CPSC page

Description
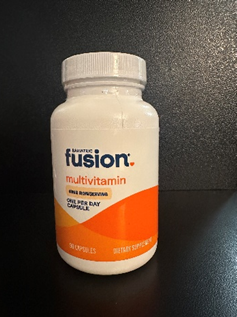
This recall involves two types of Bariatric Fusion dietary supplement bottles: high ADEK multivitamin capsules (90 and 270-count bottles) and One Per Day bariatric multivitamin capsules (90-count bottle), both with 45mg of iron. The recalled bottles are white and orange. Only bottles with smooth cap tops that lack the "push down & turn" embossed lettering are included in this recall. The Bariatric Fusion logo is printed on the front of the bottles. Lot number 0066J4, 0065J4, 0453B5 or 0370B5 is printed on the bottom of the bottles.
Hazards
Reported injuries
Remedy
Products
| Name | Model | Units |
|---|---|---|
| Bariatric Fusion Iron Multivitamin Bottles | About 4,700 |
Parties
- Manufacturer
- VitaQuest International, LLC
- Distributor
- Blueroot Health, Inc., of Middletown, Connecticut
- Made in
- United States
- Sold at
- Amazon.com, bariatricfusion.com, other websites, $57.
Consumer contact
Blueroot Health toll-free at 866-259-0602 from 8 a.m. to 6 p.m. ET Monday through Friday, email at support@bariatricfusion.com, or online at bariatricfusion.com/product-safety or www.bariatricfusion.com and click "Recall" at the bottom of the page for more information.
More images

Source: CPSC recall 25460. Inclusion in this database is not legal or medical advice.